 |
| March 26, 2024 | Volume 20 Issue 12 |
Designfax weekly eMagazine
Archives
Partners
Manufacturing Center
Product Spotlight
Modern Applications News
Metalworking Ideas For
Today's Job Shops
Tooling and Production
Strategies for large
metalworking plants
Weird science: Researchers bond meat to metal and graphite -- using electricity
Is there a way to stick hard and soft materials together without any tape, glue, or epoxy? A new study published in ACS Central Science shows that applying a small voltage to certain objects forms chemical bonds that securely link the objects together.
Reversing the direction of electron flow easily separates the two materials. This electroadhesion (EA) effect could have myriad applications in the future.
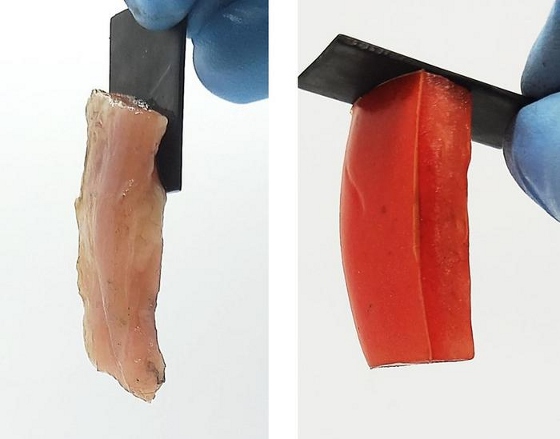
These soft materials (chicken on the left and tomato on the right) permanently stick to hard surfaces (here it is graphite) just by passing electricity through them. [Credit: Image adapted from ACS Central Science 2024, DOI:10.1021/acscentsci.3c01593]
When an adhesive is used to attach two things, it binds the surfaces either through mechanical or electrostatic forces. Sometimes, however, those attractions or bonds are difficult, if not impossible, to undo. As an alternative, reversible adhesion methods are being explored, including electroadhesion. Though the term is used to describe a few different phenomena, one definition involves running an electric current through two materials and causing them to stick together, thanks to attractions or chemical bonds.
Previously, Srinivasa Raghavan and colleagues (all at the University of Maryland) demonstrated that EA can hold soft, oppositely charged materials together, and even be used to build simple structures (see video below). This time around, they wanted to see if EA could reversibly bind a hard material, such as graphite, to a soft material, such as animal tissue.
The team first tested EA using two graphite electrodes and an acrylamide gel. A small voltage of 5 V was applied for a few minutes, causing the gel to permanently adhere to the positively charged electrode. The resulting chemical bond was so strong that, when one of the researchers tried to wrench the two pieces apart, the gel tore before it disconnected from the electrode.
Notably, when the current's direction was reversed, the graphite and gel easily separated -- and the gel instead adhered to the other electrode, which was now positively charged. Similar tests were run on a variety of materials such as metals, various gel compositions, animal tissues, and fruits and vegetables to determine the phenomenon's ubiquity.
For EA to occur, the authors found that the hard material needs to conduct electrons, and the soft material needs to contain salt ions. They hypothesize the adhesion arises from chemical bonds that form between the surfaces after an exchange of electrons. This may explain why some metals that hold onto their electrons strongly, including titanium, and some fruits that contain more sugar than salts, including grapes, failed to adhere in some situations. Metals that did adhere to soft materials in this study included tin, lead, and copper. Although not a metal, graphite also worked very well, likely due to the characteristics it shares with many metals, including being an excellent conductor of electricity. Metals that did not work in the experiments included titanium, zinc, iron, and nickel. Soft materials tested that did adhere included tomato, garlic, apple, banana, onion, potato, beef, chicken, and pork. (Is it lunch time yet?)
A final experiment demonstrated EA can also occur completely underwater, revealing an even wider range of possible applications. The team says this work could help create new batteries, enable biohybrid robotics, enhance biomedical implants, and much more.
Source: American Chemical Society
Published March 2024
Rate this article
View our terms of use and privacy policy
